open-label extension data over 4 years
WITH KEVZARA
Measures of remission do not imply drug-free remission or complete absence of disease.
DAS28-CRP=disease activity score 28-C-reactive protein; MTX=methotrexate; MTX-IR=methotrexate inadequate response; TNF-IR=tumor necrosis factor inhibitor inadequate response or intolerant; q2w=once every 2 weeks; DMARDs=disease-modifying antirheumatic drugs.
WITH KEVZARA
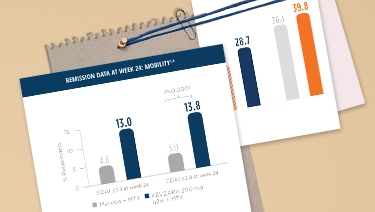
KEVZARA 200 mg + MTX SHOWED IMPROVEMENT IN CDAI REMISSION COMPARED TO PLACEBO + MTX IN MOBILITY (MTX-IR)
Measures of remission do not imply drug-free remission or complete absence of disease.
SDAI=Simple Disease Activity Index; CDAI=Clinical Disease Activity Index; MTX=methotrexate; MTX-IR=methotrexate inadequate response; TNF-IR=tumor necrosis factor inhibitor inadequate response or intolerant; q2w=once every 2 weeks; DMARDs=disease-modifying antirheumatic drugs.
INTERPRET WITH CAUTION
OVER 4 YEARS
Measures of remission do not imply drug-free remission or complete absence of disease.
SDAI=Simple Disease Activity Index; CDAI=Clinical Disease Activity Index; MTX-IR=methotrexate inadequate response; TNF-IR=tumor necrosis factor inhibitor inadequate response or intolerant; MTX=methotrexate; q2w=once every 2 weeks; DMARDs=disease-modifying antirheumatic drugs.
INTERPRET WITH CAUTION
OVER 4 YEARS
Measures of remission do not imply drug-free remission or complete absence of disease.
DAS28-CRP=disease activity score 28-C-reactive protein; MTX-IR=methotrexate inadequate response; TNF-IR=tumor necrosis factor inhibitor inadequate response or intolerant; MTX=methotrexate; q2w=once every 2 weeks; DMARDs=disease-modifying antirheumatic drugs.




